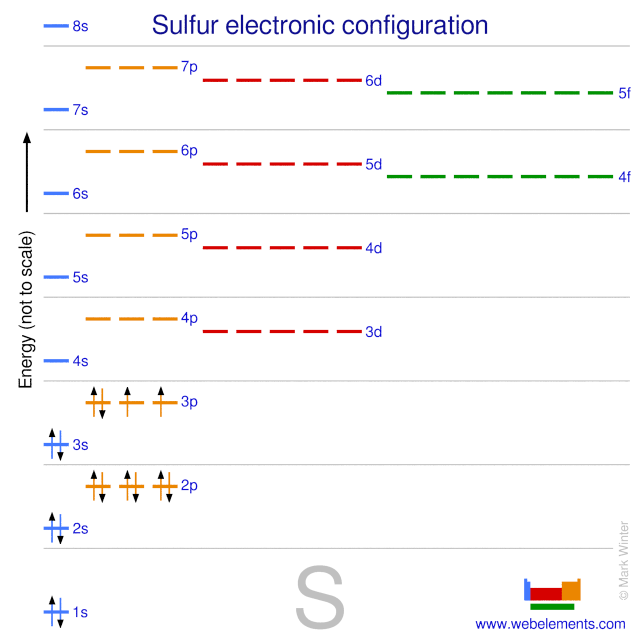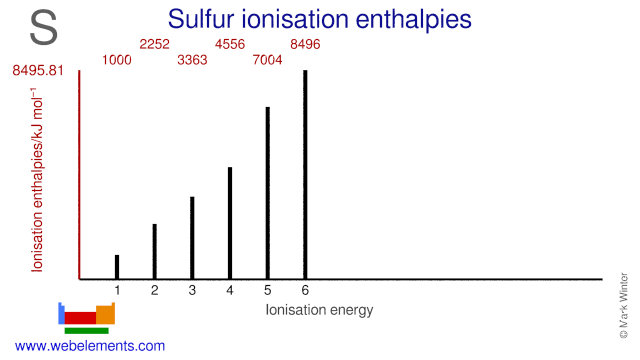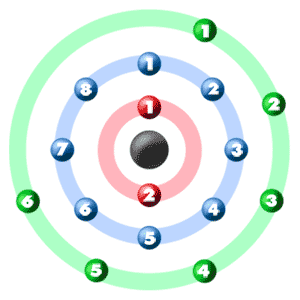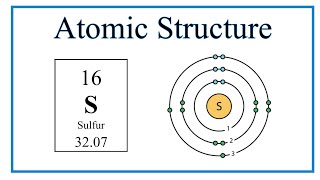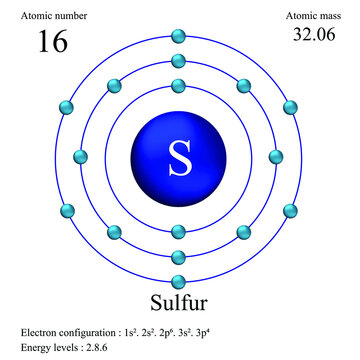
Sulfur atomic structure has atomic number, atomic mass, electron configuration and energy levels. Stock Vector | Adobe Stock
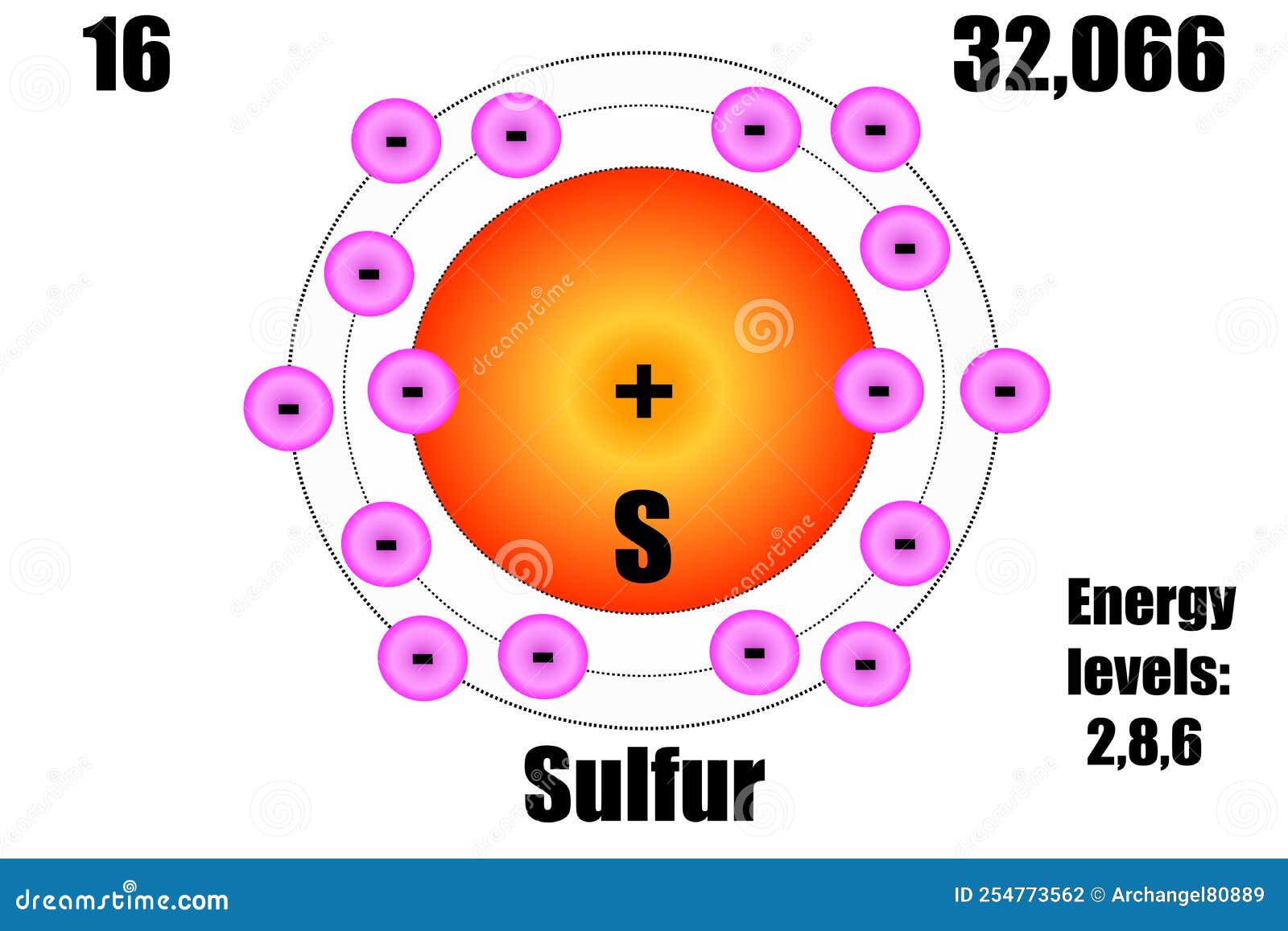
Sulfur Atom, with Mass and Energy Levels. Stock Vector - Illustration of formula, chemical: 254773562
S Sulfur Chemical Element Periodic Table. Single vector illustration, element icon with molar mass, atomic number and electron conf Stock Vector Image & Art - Alamy

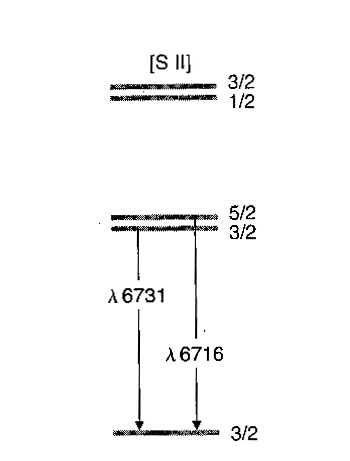
Question Video: Deducing the Energy Level Diagram Representing the Change from a Sulfur Atom to a Sulfur Ion | Nagwa

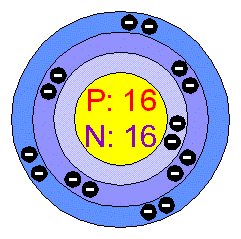
PPT - 1. How many total electrons are in a neutral atom of Sulfur ? PowerPoint Presentation - ID:2465357

A valence electron in an atom of sulfur is excited by heating a sample. The electron jumps from the s orbital to the p orbital. What is the electron configuration of the

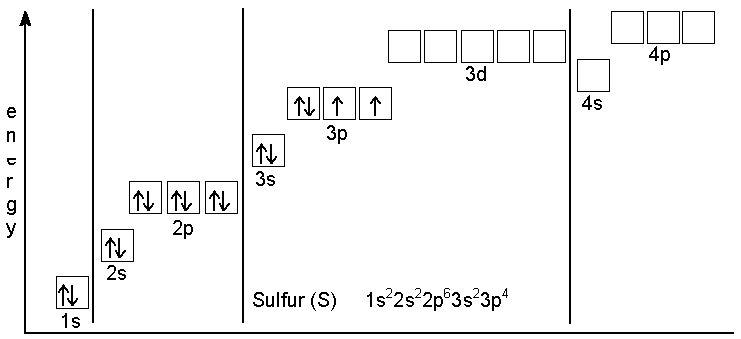
How to Write the Orbital Diagram for Sulfur (S)? | Electron configuration, Aufbau principle, Diagram

Consider a neutral atom of sulfur. There are three shells (primary quantum levels) in this atom. How many electrons does it have in: a. total b. n = 1 shell c. n =