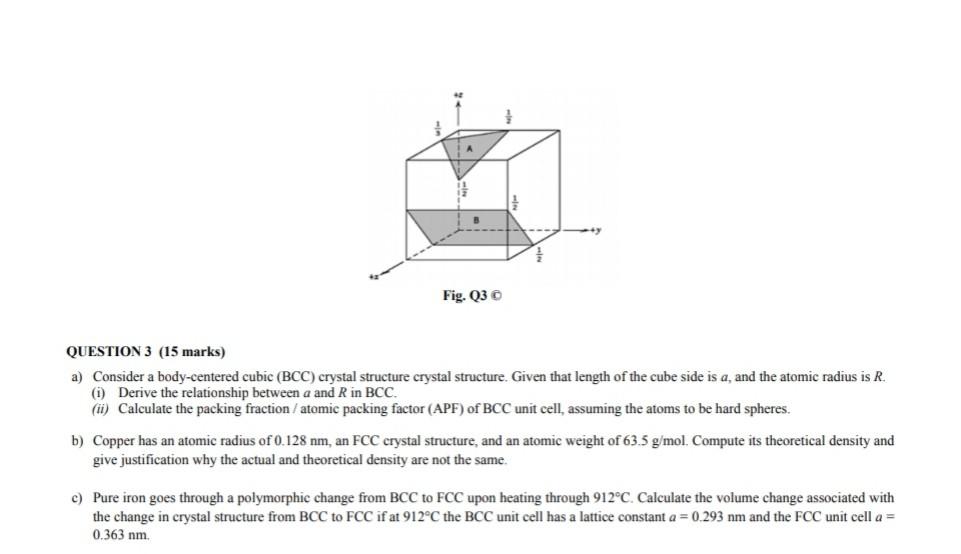
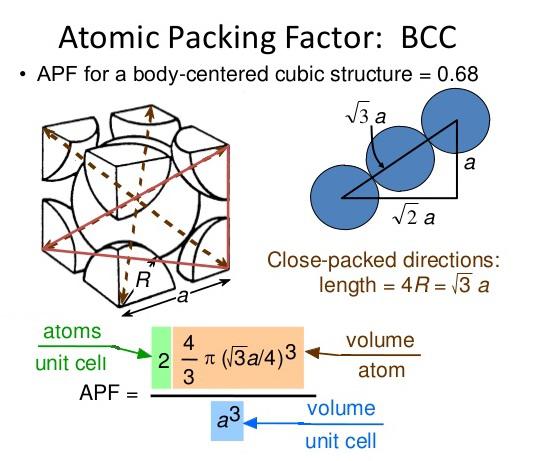
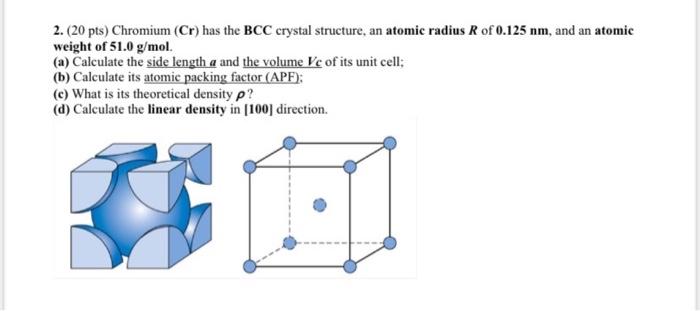
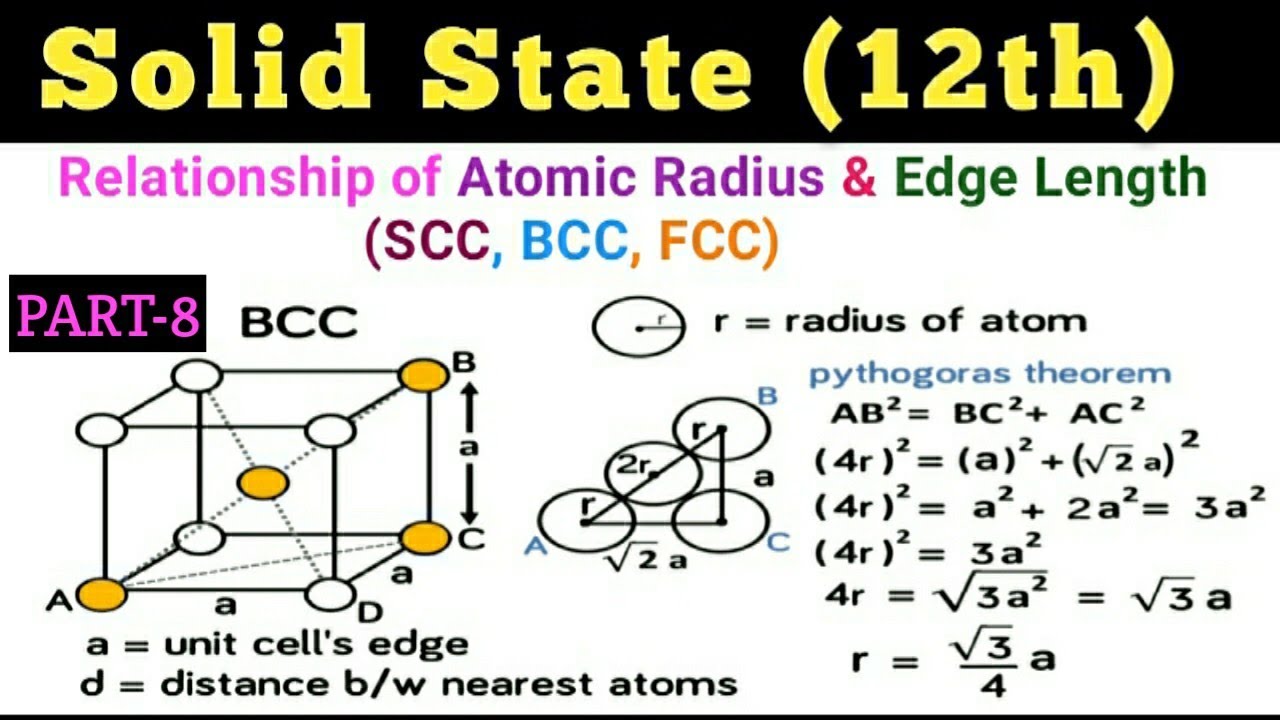
BCC, FCC structure – relation between edge length of a crystal, and atomic radius (with subtitles) - YouTube
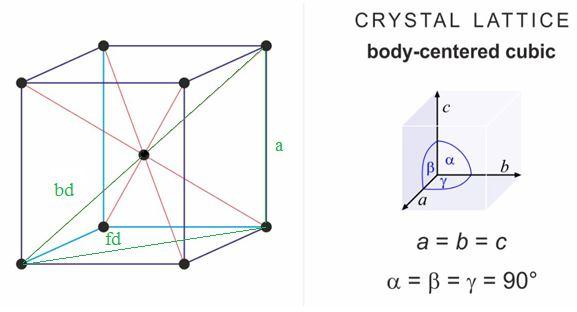
6. Atomic configuration of bcc (body centred cubic) crystal lattice and... | Download Scientific Diagram

Unit Cell Chemistry Simple Cubic, Body Centered Cubic, Face Centered Cubic Crystal Lattice Structu - YouTube
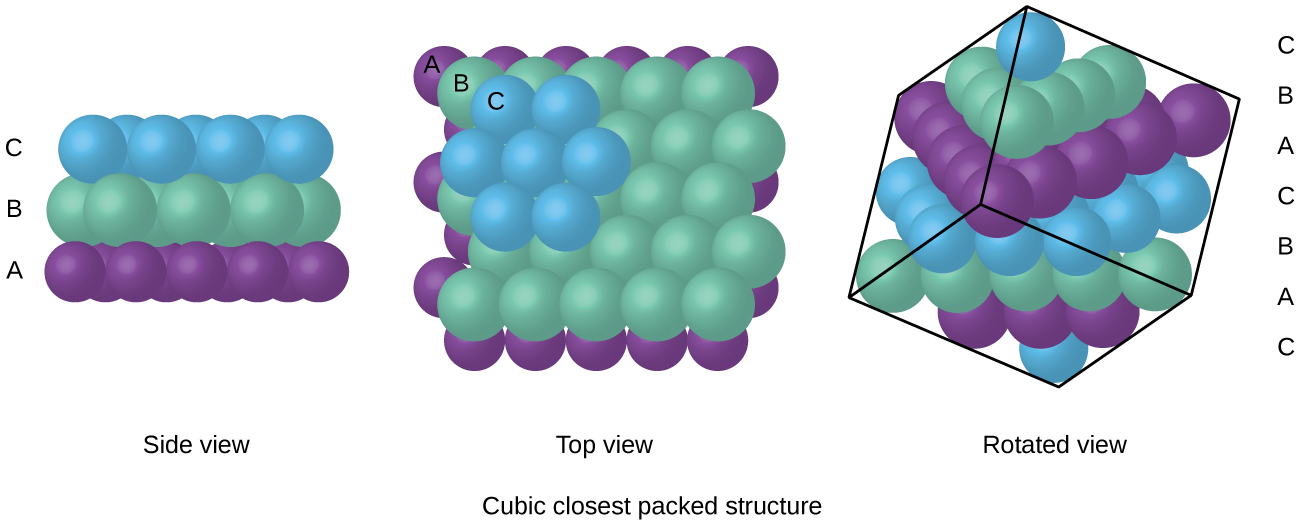
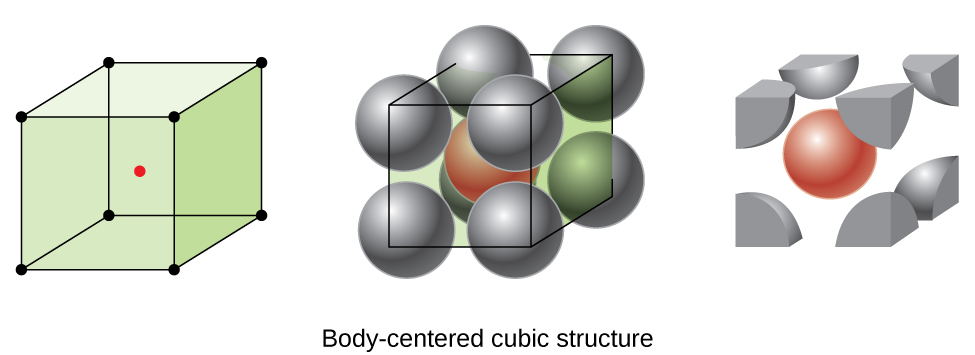
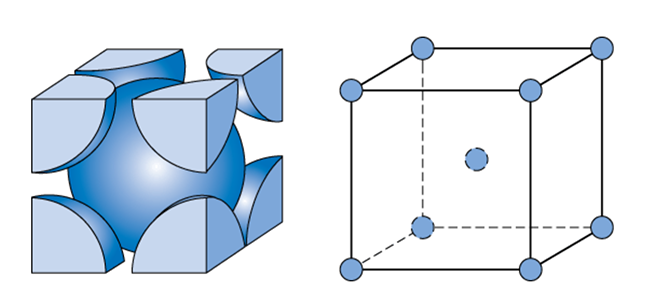
Types of Unit Cells: Body-Centered Cubic and Face-Centered Cubic (M11Q5) – UW-Madison Chemistry 103/104 Resource Book

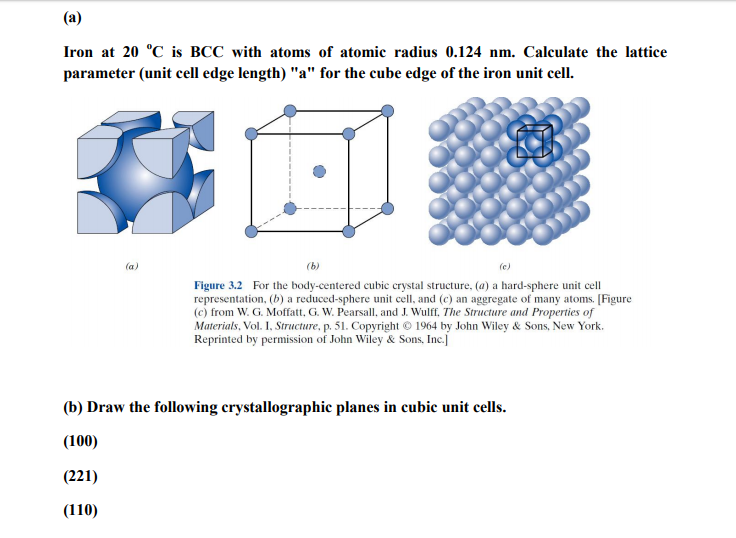
Lithium forms bcc crystals. Calculate the atomic radius of lithium if the length of the side of unit cell of lithium is 351 pm. - India Site